Abstract
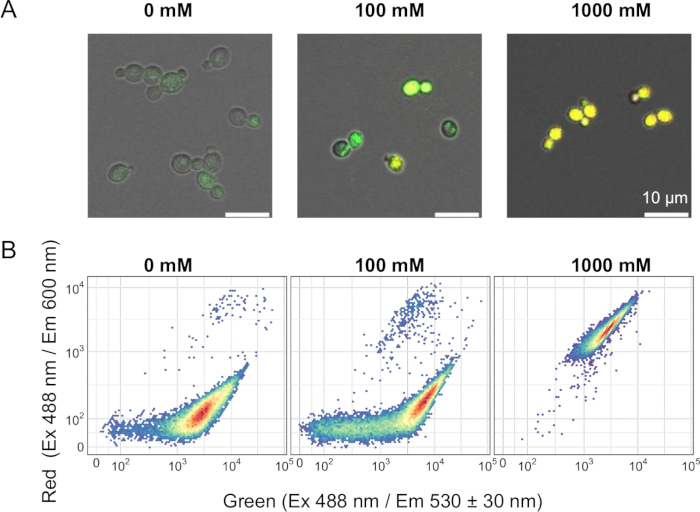
Quantifying survival is a common and critical task in yeast research. LIVE/DEAD stains based on plasma membrane integrity provide a rapid and high-throughput assay for yeast survival when coupled with flow cytometry. However, variations in staining buffer, dye concentration, incubation time, and flow cytometry settings can impact data quality and reproducibility. This protocol presents a standardized LIVE/DEAD assay for post-stress survival quantification in yeast using flow cytometry. After treating Candida glabrata, an opportunistic yeast pathogen, with different doses of hydrogen peroxide, the post-stress samples were stained with a two-component LIVE/DEAD stain consisting of SYTO 9 and propidium iodide (PI). Flow cytometry was used to distinguish live, damaged, and dead cell populations and quantify their percentages in each sample. Survival estimates based on the percent live statistic were compared to the Colony Forming Unit (CFU) result on the same sample. The two methods yielded consistent results for the mock- and lethal dose (1 M H2O2)-treated samples. At the sublethal dose of 100 mM H2O2, SYTO 9/PI estimated a higher survival rate than CFU, reflecting a key difference between the two, where the protocol presented here evaluates cell survival immediately after the stress, while CFU quantifies the percent of cells able to recover and reproduce. Hence, this protocol measures viability at an earlier stage of the cell-death process. In summary, the protocol described here provides a fast and scalable alternative to CFU for post-stress survival quantification in yeast. Its results provide complementary information to CFU by evaluating survival at an earlier stage and distinguishing between dead and damaged cells.