Abstract
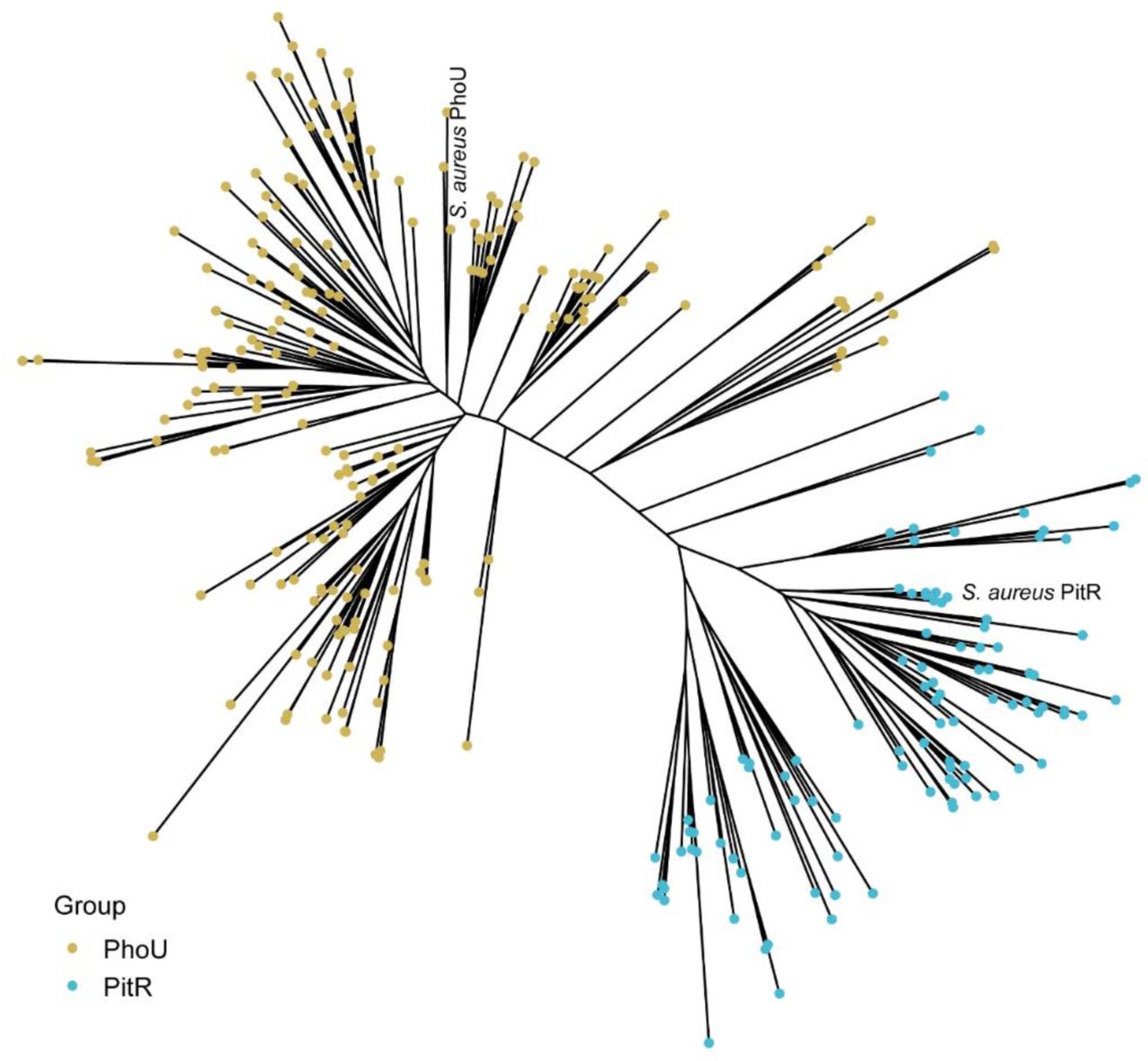
Phosphate is both essential for life and toxic, necessitating the tight regulation of its acquisition. Based on Escherichia coli, most bacteria are thought to use a single accessory protein that monitors import to regulate phosphate homeostasis. This work reveals that many bacteria possess multiple, distinct families of accessory regulators, with each family regulating homeostasis in conjunction with a unique importer family. The antibiotic-resistant pathogen Staphylococcus aureus can obtain phosphate from divergent environments and possesses accessory-transporter pairs from all three identified groups. Investigations with S. aureus revealed that all three accessory proteins can regulate phosphate homeostasis, but that there is a hierarchy, which is dictated by the environment. Multiple accessory regulators are independently necessary for S. aureus to cause infection. Thus, microbes possess not one, but multiple distinct groups of accessory regulatory proteins, and this diversity enables them to control phosphate homeostasis across environments, including those encountered during infection. IMPORTANCE Phosphate homeostasis is critical for bacterial survival, but this nutrient is simultaneously vital and harmful in excess. The prevailing model based on Escherichia coli proposed that bacteria monitor phosphate import through a sole accessory regulatory protein. This work both challenges and extends this paradigm by demonstrating that bacteria frequently possess multiple evolutionarily distinct accessory regulator families, with each tightly linked to a specific transporter. Using the versatile pathogen Staphylococcus aureus, this work demonstrates that these regulatory proteins function in an environment-dependent hierarchy to control phosphate balance. During host infection, multiple accessory regulators are independently necessary. This regulatory expansion represents a fundamental strategy by which bacteria adapt phosphate acquisition to their surroundings, particularly within a dynamic host environment.